Research Question
|
The purpose of this research is to see the effects of adding water to the reline solution, choline chloride and urea in a 1:2 molar ratio, and analyzing how that affects the size of gold nanoparticles when the gold salt HAuCl4 is mixed with NaBH4(a reducing agent).
|
Uses of Gold Nanoparticles |
Hypothesis |
|
Gold in its ordinary bulk form is considered to be a noble metal because it is mostly unreactive with other atoms and molecules. When gold is in its extremely small form the nanoparticles have a variety of implications in the modern world. Gold nanoparticles can be used to reduce the emissions of harmful gasses. For example, it can be used to convert carbon monoxide into carbon dioxide, making it a plausible chemical to replace platinum in catalytic converters in cars. These nanoparticles can also be manipulated to assist cancer detection.
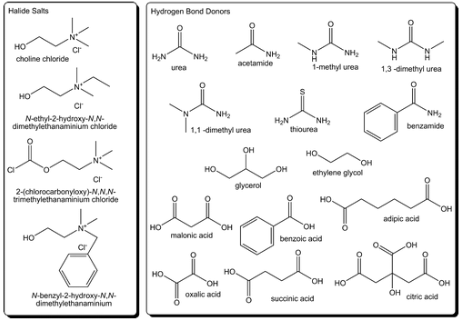
Deep Eutectic SolventsDeep eutectic solvents are composed of a halide salt mixed with a hydrogen bond donor and together they form an ionic liquid. These solvents are very effective at disassociating other molecules because of their strong intermolecular forces, but these strong intermolecular forces also cause them to be very viscous, which slows down the reaction. Deep eutectic solvents are also very cheap to make so they have many industrial applications to make gold nanoparticles.
|
It is hypothesized that increasing the concentration of water in the reline solution will increase the average size of the gold nanoparticles. This is because the gold would not stay in solution as easily as it would in higher concentrations of reline. This would able the nanoparticles to clump up and make bigger particles.
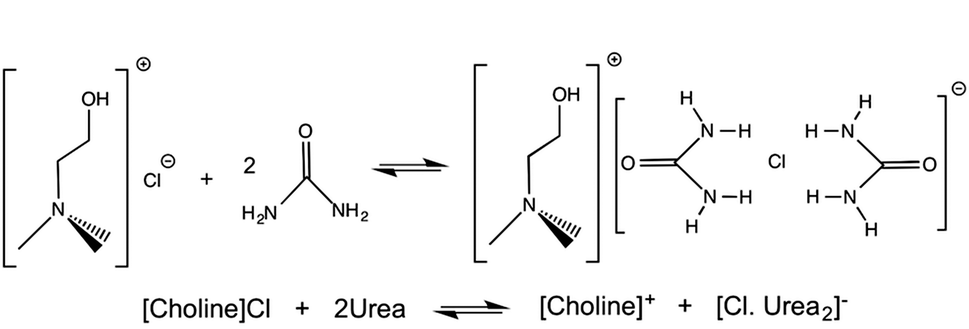
RelineIn this research the deep eutectic solvent being used is known as reline. This is made through mixing choline chloride and urea in a one to two ratio respectively. Reline is an ionic liquid because it is made from two ionic salts, and it changes from two solids into a viscous liquid.
|